Enasidenib is an oral targeted therapy indicated for relapsed or refractory acute myeloid leukemia with IDH2 mutations.
I. What is Enasidenib?
1. Drug Profile
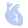
(1) Enasidenib (brand name: IDHIFA) is an isocitrate dehydrogenase-2 (IDH2) inhibitor. It was first approved in the United States in 2017 for the treatment of adult patients with relapsed or refractory acute myeloid leukemia (AML) harboring an IDH2 mutation.
(2) IDH2 mutation status must be confirmed using an FDA-approved companion diagnostic test prior to treatment initiation.
2. Dosage Form and Administration
(1) It is available as tablets in two strengths: 50 mg and 100 mg.
(2) The recommended dosage is 100 mg orally once daily, with or without food. Tablets should be swallowed whole; do not chew, split, or crush them.
(3) Treatment should be continued until disease progression or unacceptable toxicity occurs. In the absence of these, treatment should be administered for at least 6 months to allow for a clinical response.
II. Mechanism of Action and Drug Interactions
1. Mechanism of Action
(1) Enasidenib selectively inhibits the mutant IDH2 enzyme (including variants such as R140Q, R172S, and R172K), with an inhibitory potency approximately 40-fold greater than that against the wild-type enzyme.
(2) By inhibiting mutant IDH2, it reduces the level of the oncogenic metabolite 2-hydroxyglutarate, thereby inducing myeloid cell differentiation, decreasing blast counts, and increasing the proportion of mature myeloid cells.
2. Effects on Hepatic Enzymes
Enasidenib is an inhibitor of CYP1A2 and CYP2C19, as well as an inducer of CYP3A.
(1) CYP1A2 substrates (e.g., caffeine): Exposure increases by 655%. Reduced frequency of caffeine intake is recommended.
(2) CYP2C19 substrates (e.g., omeprazole): Exposure increases by 86%. Concomitant use should be avoided or dosage adjusted.
(3) CYP3A substrates (e.g., midazolam, some antifungal agents): Exposure decreases by 43%, which may lead to loss of antifungal efficacy. It also reduces concentrations of hormonal contraceptives, requiring a switch to non-hormonal contraceptive methods.
3. Effects on Transporters
(1) Enasidenib inhibits OATP1B1, OATP1B3, BCRP, and P-glycoprotein.
(2) OATP1B1/1B3/BCRP substrates (e.g., rosuvastatin): Exposure increases by 244%–366%, potentially raising the risk of toxicity.
(3) P-glycoprotein substrates (e.g., digoxin): Exposure increases by 20%–26%, requiring enhanced monitoring.
III. Management of Missed Doses and Overdose
1. Missed Dose Management
(1) If a dose is missed or vomiting occurs after administration, the dose should be taken as soon as possible on the same day, and the normal dosing schedule resumed the following day.
(2) Do not take two doses at one time to make up for a missed dose.
2. Overdose Management
(1) In the event of overdose, closely monitor the patient’s vital signs and adverse reactions (especially differentiation syndrome, abnormal hepatic and renal function, etc.), and provide supportive care based on clinical manifestations.
(2) Immediate contact with a physician is recommended.
3. Importance of Regular Administration
(1) Given enasidenib’s long half-life of 7.9 days, steady-state plasma concentrations are reached in approximately 29 days with an approximately 10-fold accumulation. Therefore, consistent daily dosing at the same time is critical to maintaining stable therapeutic efficacy.
(2) Setting an alarm reminder is recommended to avoid missed doses.

Free Inquiry