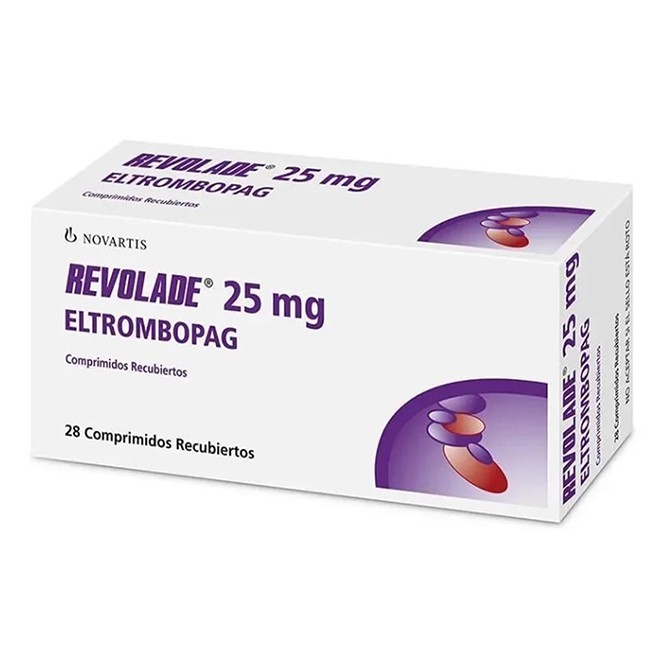
Eltrombopag is a targeted drug used to increase platelet count. Patients are most concerned about its price, medical insurance reimbursement, and actual therapeutic effects.
I. Is Eltrombopag Covered by Medical Insurance?
It has been included in the National Medical Insurance Catalogue.
Eltrombopag was added to the Category B of the National Medical Insurance Catalogue in 2017 for the treatment of chronic immune thrombocytopenia (ITP) refractory to previous therapies and severe aplastic anemia (SAA).
Medical insurance reimbursement is subject to the following conditions: patients must be diagnosed by a physician with eligible indications and obtain a prescription issued by a hospital.
Reimbursement ratios vary by region. Employee medical insurance generally covers 50%–80% of the cost, significantly reducing patients’ out-of-pocket expenses.
For specific reimbursement policies, it is recommended to consult the local medical insurance bureau or the pharmacy of the treating hospital.
II. How Much Does Eltrombopag Cost?
Price before medical insurance reimbursement:
A box of 25 mg × 28 tablets costs approximately 6,000–6,500 RMB.
A box of 50 mg × 28 tablets costs approximately 10,000–11,000 RMB.
Reference price of overseas originator brand:
The imported originator brand Revolade (Novartis) without medical insurance coverage is relatively expensive in the U.S. market, with a 50 mg × 30-tablet pack costing about 5,500–6,500 USD.
Status of generic drugs:
Generic versions of eltrombopag have been approved in China, priced lower than the originator brand. Selection must be made under the guidance of a physician.
III. What Are the Therapeutic Effects of Eltrombopag?
1. Efficacy in Adult ITP Patients
(1) According to the RAINSE clinical trial data in the FDA label, 60% of patients achieved a sustained platelet response (platelet count ≥ 50×10⁹/L and ≤ 400×10⁹/L for more than 6 consecutive weeks) at 6 months of treatment, compared with only 10% in the placebo group.
(2) The mean duration of maintained target platelet levels was 11.3 weeks, versus only 2.4 weeks in the placebo group.
(3) Proportion of patients requiring emergency rescue treatment: 18% in the eltrombopag group and 40% in the placebo group.
2. Efficacy in Pediatric ITP Patients
(1) In the PETIT2 study, 41% of patients aged 1–17 years met the primary endpoint (platelet count ≥ 50×10⁹/L for at least 6 weeks during weeks 5–12 of treatment), compared with only 3% in the placebo group.
(2) 75% of pediatric patients in the eltrombopag group achieved target platelet levels at least once, versus only 21% in the placebo group.
3. Efficacy in Treatment-Naïve Severe Aplastic Anemia (SAA)
After 6 months of combination therapy with standard immunosuppressive therapy, the overall response rate reached 79%, including a complete response rate of 44%. The median duration of overall response exceeded 24 months.
4. Efficacy in Refractory SAA
For patients unresponsive to immunosuppressive therapy, the overall response rate to eltrombopag treatment was 40%. The median duration of response has not been reached (most patients maintained a sustained response).
5. Onset of Action
In ITP patients, platelet count generally begins to rise within 1–2 weeks of administration, with peak efficacy achieved at approximately 2 weeks. Platelet count returns to baseline levels about 1–2 weeks after drug discontinuation.
IV. Important Precautions
1. Medical Insurance Reimbursement Conditions
(1) Eligibility is restricted to the approved payment scope, generally requiring a platelet count below 30×10⁹/L and a risk of bleeding.
(2) Pre-registration is required before reimbursement.
2. Administration Reminders
(1) Take on an empty stomach (1 hour before meals or 2 hours after meals). Avoid concomitant intake with foods or medications containing calcium, iron, or magnesium (allow at least 4 hours between doses).
(2) Liver function and platelet count must be monitored regularly during treatment.
3. Contraindicated Population
Eltrombopag is contraindicated in patients with myelodysplastic syndromes (MDS), as it may increase the risk of progression to acute myeloid leukemia.

Free Inquiry